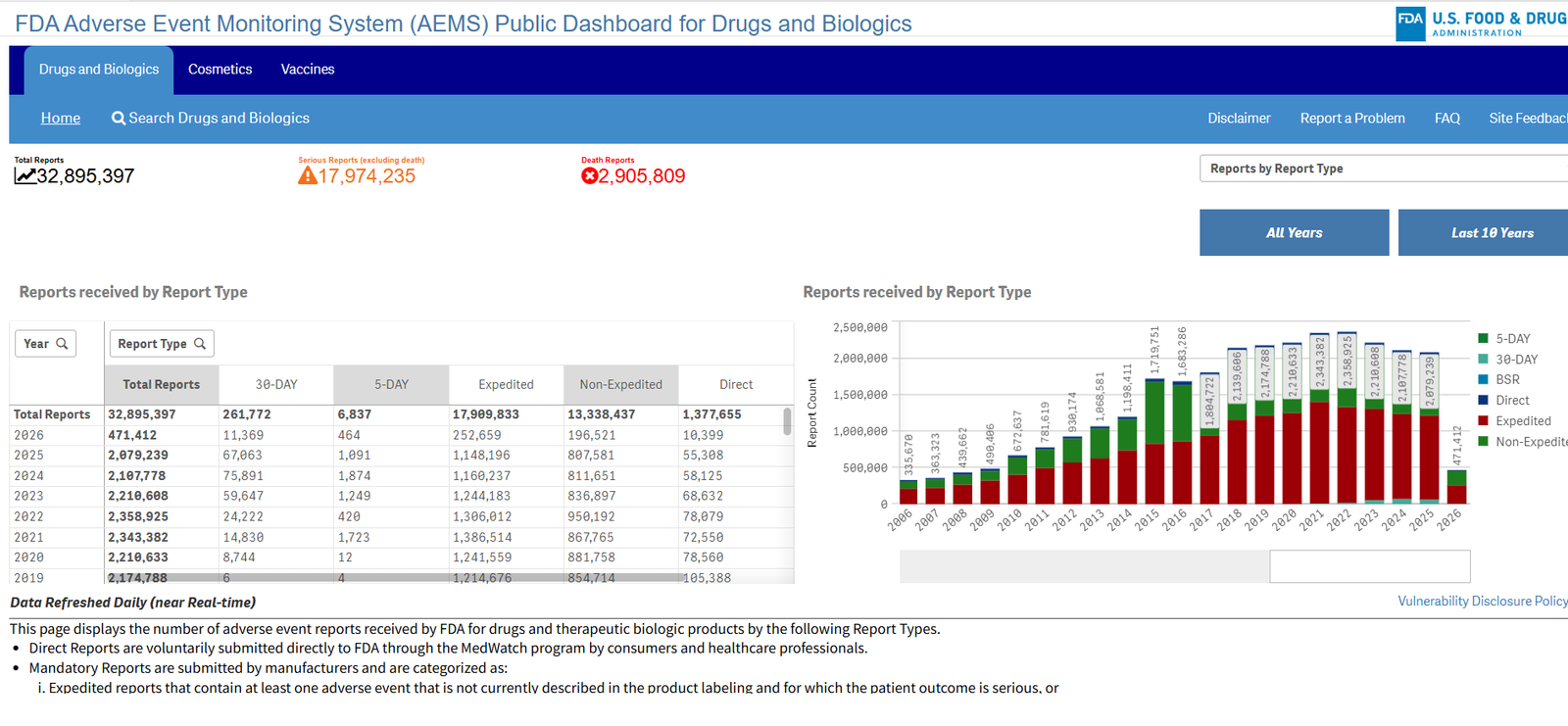
On March 11, 2026, the U.S. Food and Drug Administration (FDA) officially announced the launch of the Adverse Event Monitoring System (AEMS). This brand-new unified platform and interactive public data dashboard is designed to analyze and process adverse event reports with greater efficiency.
The system is scheduled to be fully operational by the end of May 2026. This transition will bring significant changes to the industry landscape and the way the public accesses data. Here is the core information you need to know:
A “Unified” Era: Covering All Regulated Products
The launch of AEMS marks a major milestone in the FDA’s data integration efforts. It will provide real-time adverse event reporting across all FDA-regulated product categories, including:
- Medical Products and Devices
- Vaccines
- Tobacco Products
- Food and Cosmetics
- Veterinary Medicines
Retiring Legacy Systems
To achieve this integration, the FDA will decommission several aging, decentralized reporting systems across various centers. The new AEMS will directly replace the following well-known platforms:
- FAERS: Formerly used for reporting on drugs, biological products, cosmetics, and color additives.
- VAERS: Formerly used for vaccine-related incidents.
- AERS: Formerly used for animal drugs and animal food.
By consolidating these systems into a single platform, the FDA aims to streamline the reporting process and enhance cross-product regulatory oversight.
Efficiency and Cost Savings: $120 Million Saved and Expanded Functionality
Beyond serving as a core database for adverse events, AEMS will expand its operational scope to become a centralized management hub. It will unify the processing of consumer complaints, regulatory violation reports, and whistleblower submissions from different FDA centers.
This initiative offers two major advantages:
- Massive Cost Savings: The FDA estimates that by consolidating functions and shutting down legacy systems, approximately $120 million in operating costs will be saved over the next five years.
- Increased Transparency: Previously, the public could only access data on a quarterly basis. Now, the AEMS public dashboard will enable near real-time data publishing. The FDA expects this to significantly reduce the volume of Freedom of Information Act (FOIA) requests for data.
Usage and Important “Disclaimers”
With the rollout of the FDA AEMS public dashboard, the general public, healthcare providers, and industry professionals will have seamless access to search for adverse event information.
Critical Reminder (Official FDA Limitations Statement) While data access has become easier, the FDA emphasizes several key limitations when interpreting this data:
- No Causality Implied: The presence of an adverse event report in the system does not mean the product “caused” the event.
- Unverified Data: Information submitted to the system has not been independently verified by the FDA.
- Potential Data Flaws: The database may contain duplicate submissions or incomplete reports.
- Inability to Calculate Incidence: This data serves as a reference for monitoring only and cannot be used to infer or establish the exact incidence rate of adverse events.
The introduction of AEMS is a significant milestone in the FDA’s journey toward digital transformation and data transparency. For pharmaceutical companies, R&D institutions, and the public, more timely data means faster response times and stronger risk management capabilities. We will continue to monitor the system’s performance following its full implementation in late May!